Hydrogen
| Isotope | Atomic mass (Da) | Isotopic abundance (amount fraction) |
|---|---|---|
| 1H | 1.007 825 0322(6) | [0.999 72, 0.999 99] |
| 2H | 2.014 101 7781(8) | [0.000 01, 0.000 28] |
The major stable isotope ¹H is also known as protium. The minor stable isotope ²H is known as deuterium, with symbol D. The low concentration of ²H in normal sources of hydrogen may have delayed its discovery until 1931 when hydrogen isotope fractionation was demonstrated by distillation, electrolysis, evaporation, and in environmental samples. The atomic weight of hydrogen has been based on mass-spectrometric measurements since 1938. In its report for 1961, CIAAW recommended Ar(H) = 1.007 97(1) based on the average and the range of ²H concentrations measured in hydrogen extracted from fresh- and saltwaters; however, it was noted that substances other than water could have a wider range of atomic weights.
The currently accepted best measurement of the isotopic composition of hydrogen from a single natural source was performed on VSMOW (distributed by the IAEA and NIST), the reference material endorsed by CIAAW as the basis of the delta scale for relative isotope-ratio measurements. According to this measurement, VSMOW has an amount fraction of x(²H) = 0.000 155 74(5), corresponding to Ar(H)VSMOW = 1.007 981 75(5). The uncertainty of that value corresponds to a δ(²H) uncertainty of 0.3 ‰, which is equal to or slightly smaller than typical uncertainties of most relative isotope-ratio measurements of H. Variations in the isotopic composition of hydrogen in chemicals and natural terrestrial systems are known to exceed +1000 ‰, which is much larger than the uncertainty due to isotope-ratio measurements.
For water sources, the range of published δ(²H) values extends from −495 ‰ (Ar(H) = 1.007 9042), to +129 ‰ (Ar(H) = 1.008 0020). Seawater, the largest reservoir of water near the Earth's surface, has a relatively uniform isotopic composition and atomic weight near that of VSMOW; whereas precipitation, polar ice, lakes, rivers, and groundwaters have atomic weights that range widely, generally decreasing with latitude and elevation and increasing with evaporation. The highest δ(²H) value reported for a material of natural terrestrial origin is +180 ‰ for atmospheric H2. An unusual anthropogenic occurrence of water from a H2S well yielded δ(²H) values as high as +375 ‰ (Ar(H) = 1.008 0404). Hydrogen samples with low atomic weights, some of which are outside the range of the standard atomic-weight uncertainties, have been reported from some types of natural and artificial H2 gases, hence the annotation "g". The naturally occurring hydrogen sample with the lowest atomic weight, δ(2H) = −836 ‰ and Ar(H) = 1.007 8507, is H2 gas collected from a natural gas well in Kansas, USA. That sample, and other similar ²H-depleted H2-rich natural gases elsewhere, may have formed by natural chemical reduction of water during low-temperature reactions with ultramafic (Fe-Mg-silicate) rocks. Hydrogen gases produced artificially by electrolysis and as by-products of petrochemical processing commonly are depleted in ²H. Though not natural, those gases are considered to be important because they are used commonly in laboratories. Commercial tank H2 has been reported to have δ(²H) as low as −813 ‰ (Ar(H) = 1.007 8543).
The radioactive isotope ³H, also known as tritium with symbol T, decays by negative beta emission to ³He with a half-life of 12.3 years. Tritium is formed naturally in the atmosphere by cosmic-ray reactions such as 14N(n,t)12C and artificially in nuclear reactors. Large quantities of tritium were injected into the atmosphere as a by-product of thermonuclear bomb tests, mostly in the 1950s and 1960s. Tritium reacts in the atmosphere to form HTO and other compounds that are distributed with widely varying concentrations in the near-surface environment of the earth. Those variations, and other local anthropogenic ³H anomalies, are used commonly in environmental studies; however, concentrations of tritium in normal sources of hydrogen are too low by several orders of magnitude to have a measurable effect on the atomic weight of hydrogen.
Atomic weights of the elements 2009 by M.E. Wieser and T.B. Coplen. Pure Appl. Chem. 2011 (83) 359-396
CIAAW
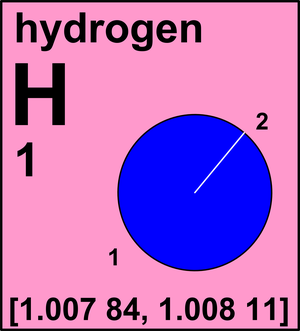
Hydrogen
Ar(H) = [1.007 84, 1.008 11] since 2009
The name derives from the Greek hydro for "water" and genes for "forming" because it burned in air
to form water. Hydrogen was discovered by the English physicist Henry Cavendish in 1766.
Natural variations of hydrogen isotopic composition
Isotopic reference materials of hydrogen.